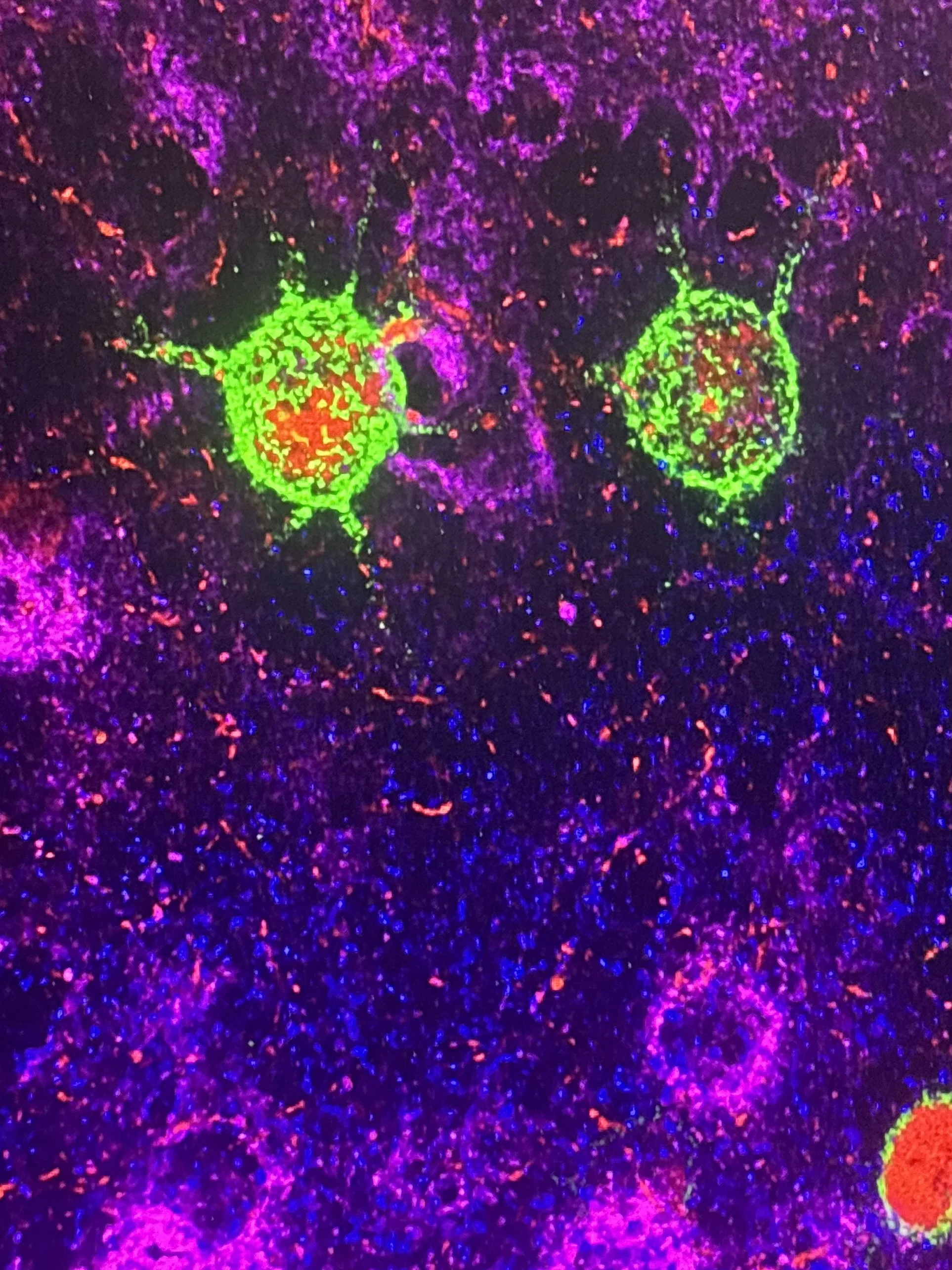
Role of Insular Perineuronal Nets in Alcohol Use Disorder
Binge and aversion-resistant alcohol consumption models of alcohol use disorder have been shown to be regulated by a subset of the extracellular matrix called perineuronal nets (PNNs). These structures, composed of a hyaluronan backbone with associated chondroitin sulfate proteoglycans (CSPGs) and link proteins, have been thought to stabilize synapses and therefore maintain synaptic memories. In models of abuse, up- or down-regulation of these nets allow the brain to encode drug-associated memories that may worsen disease pathology. In our lab’s model of binge alcohol consumption, we have shown that PNNs are upregulated in the anterior insular cortex, a region associated with risk evaluation. In addition, enzymatic degradation of anterior insular PNNs decreases aversion-resistant alcohol consumption. Our current work focuses on how drugs or pharmacotherapies influence PNN abundance and alcohol consumption, along with understanding the biological mechanisms behind these changes.
Transcriptional and Epigenetic Control of Alcohol Use Disorder
Alcohol use disorder (AUD) leads to long-lasting changes in brain circuits that promote compulsive drinking and relapse. These persistent adaptations arise from coordinated changes in gene expression and cellular plasticity across multiple brain cell types. Our research explores how transcriptional and epigenetic regulators shape alcohol-induced brain plasticity. We are particularly interested in astrocytes, which actively modulate the synaptic environment, structural stability, and circuit remodeling following alcohol exposure. We investigate how alcohol reshapes cell-type-specific gene expression programs in brain regions such as the prefrontal cortex and hippocampus, and how these changes influence structural plasticity, extracellular matrix dynamics and behavioral outcomes. By combining conditional genetic models with transcriptomic and epigenomic approaches, we aim to define the molecular mechanisms that drive maladaptive plasticity in alcohol use disorder. Understanding how gene regulatory pathways shape circuit stability and flexibility may reveal new strategies to promote resilience and reduce relapse vulnerability.
Role of Estrogen in Alcohol Use Disorder
Rodent research on drugs of abuse has traditionally been understudied in females due to the impact of estrogen, the main female reproductive hormone. Estradiol, the most abundant estrogen subtype, binds to estrogen receptors in the nucleus, which act as transcription factors to drive gene expression. Our lab has shown that estradiol levels, as seen in different stages of the estrous cycle, influence perineuronal net abundance and intensity. In addition, many behavioral and drinking-related behaviors we have investigated have revealed sex differences, implicating estrogen as a mediator of transcriptional changes. Our current work aims to reveal mechanisms by which estrogen is influencing alcohol-related behaviors and gene expression.